ECTRON the first choice for Neurological Rehabilitation Technology
Ectron are trusted by the world's leading technology companies to supply their devices to the UK market and have an enviable network of national and international contacts.
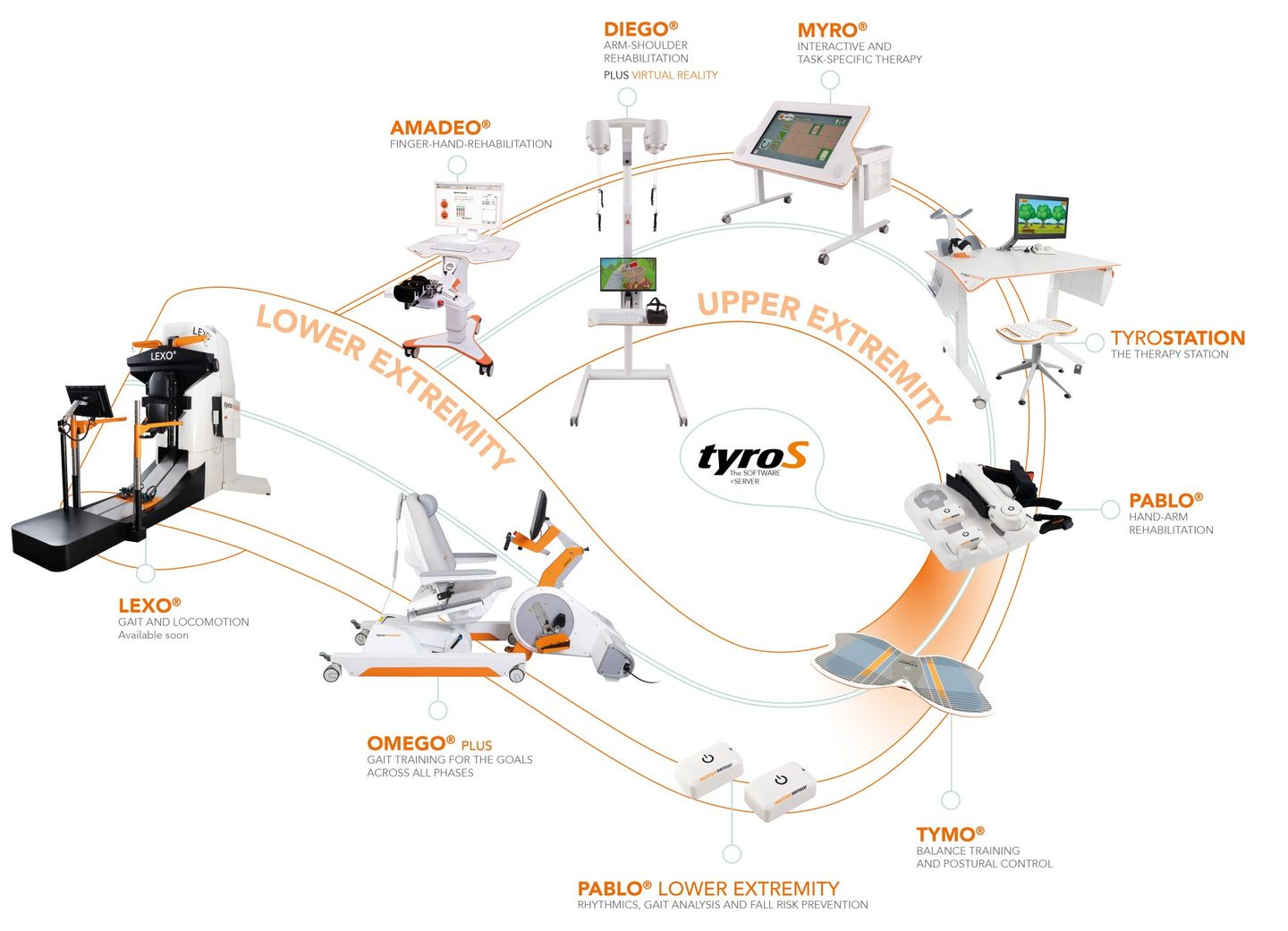
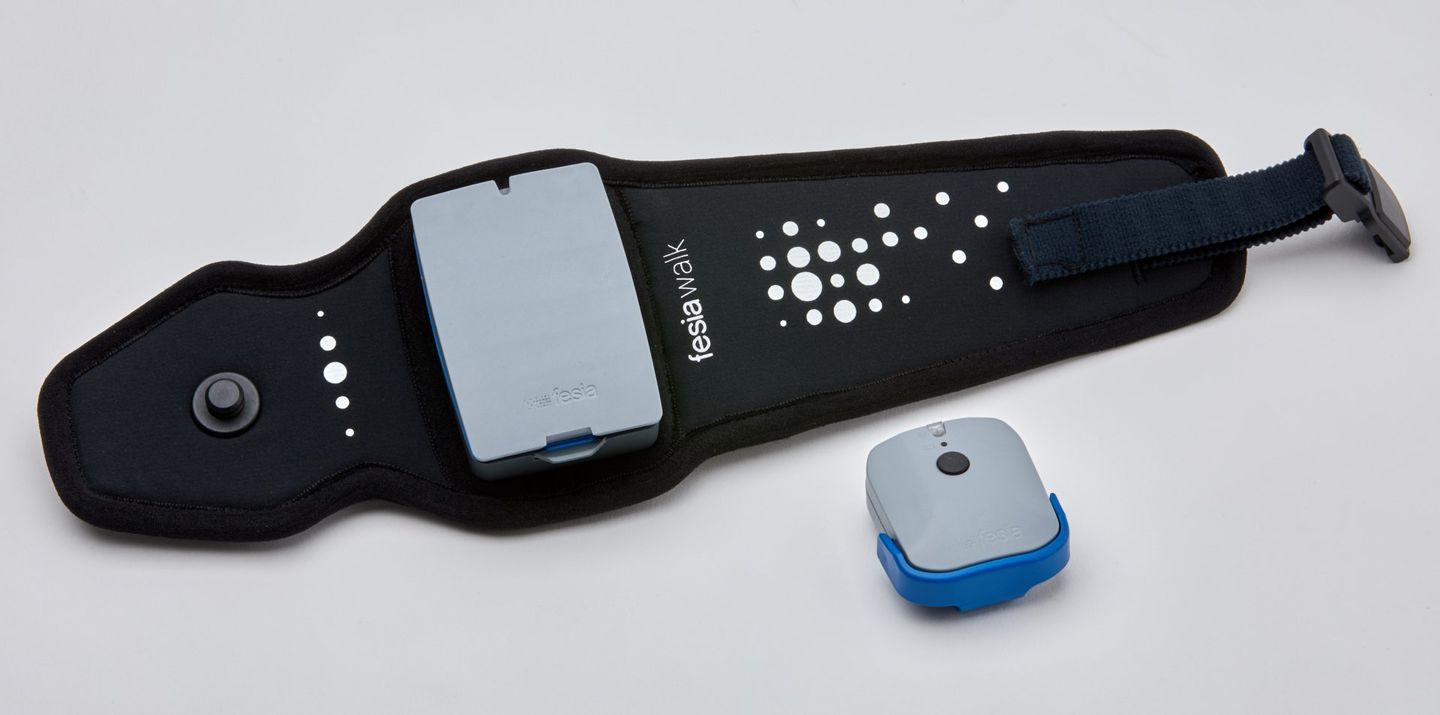
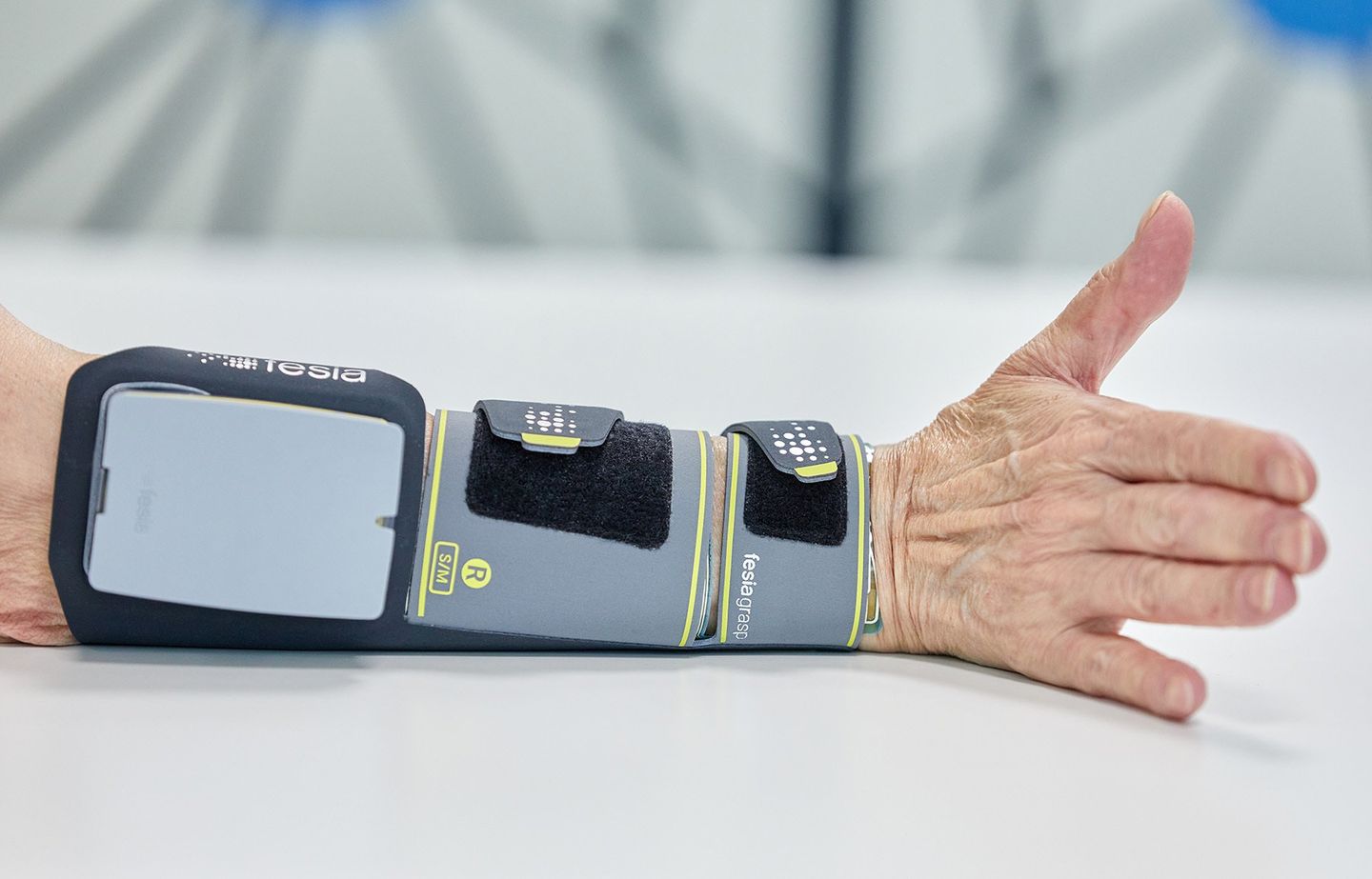
For the last ten years Ectron has been recognised as the UK leader in the implementation of neurological rehabilitation robotic and sensor-based technology clinics solutions. We are proud to have worked with some of the UK's leading and pioneering Neurological Rehabilitation Centres including the Hospital of Neurology and Neurosurgery, Stoke Mandeville Spinal Injury Unit, MOTIONrehab's Intensive Neurological Rehabilitation Centres & Hobbs Rehabilitation. Ectron can advise you on the right neurological rehabilitation solution for your clinic or hospital.
THE HISTORY OF ECTRON
Ectron was founded in 1950 by the late Dr. Russell to manufacture medical equipment for the use within psychiatry and went on to become the world’s leading manufacturer of Electro Convulsive Therapy (ECT) devices. Ectron has since expanded its range of products and is now working with international partners in new exciting areas.
Ectron's key business is to supply the latest medical devices for use in neurological rehabilitation, psychiatry, neurology and psychology. The company prides itself on introducing new and emerging technologies along with supplying established products, focusing on treatment and rehabilitation of neurological injuries and illness.